With patient welfare dangling in the balance, disruptive innovation is a permanent apprehension and product failure is not an option in healthcare. Implementing engineering simulation throughout development can make all the difference in introducing new treatments to patients and product deadlines.
When you build reliability into the design process, it advances compliance with tightening FDA regulations and saves lives. Since clinical testing can’t tackle every possible situation, medical device companies embrace engineering simulation to systematically recognize and reconcile worst-case scenarios.
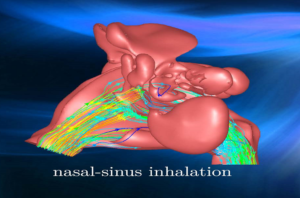
With patient welfare dangling in the balance, disruptive innovation is a permanent apprehension and product failure is not an option in healthcare. Implementing engineering simulation throughout development can make all the difference in introducing new treatments to patients and product deadlines.
When you build reliability into the design process, it advances compliance with tightening FDA regulations and saves lives. Since clinical testing can’t tackle every possible situation, medical device companies embrace engineering simulation to systematically recognize and reconcile worst-case scenarios.

More About Healthcare Applications
- Implantable cardiovascular devices — stents, coils, heart valves and pacemakers — are complex as a result of exacting product and regulatory (FDA) specifications. The study of hemodynamics is critical to cardiac device engineering, which can benefit greatly from engineering simulation and advanced fluid–structure interaction modeling.
- Globalization addresses the lack of engineering resources in non-industrialized countries. It also enables the healthcare industry to customize global concepts (device, product or process) to meet local needs.





